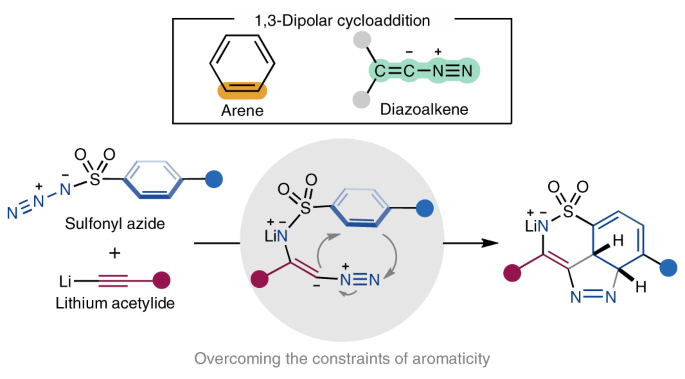
Arenes participate in 1,3-dipolar cycloaddition with in situ-generated diazoalkenes

Chemistry of Compounds Based on 1,2,3-Triazolylidene-Type

決め手はジアゾアルケン!!芳香環の分子内1,3-双極子付加環化反応
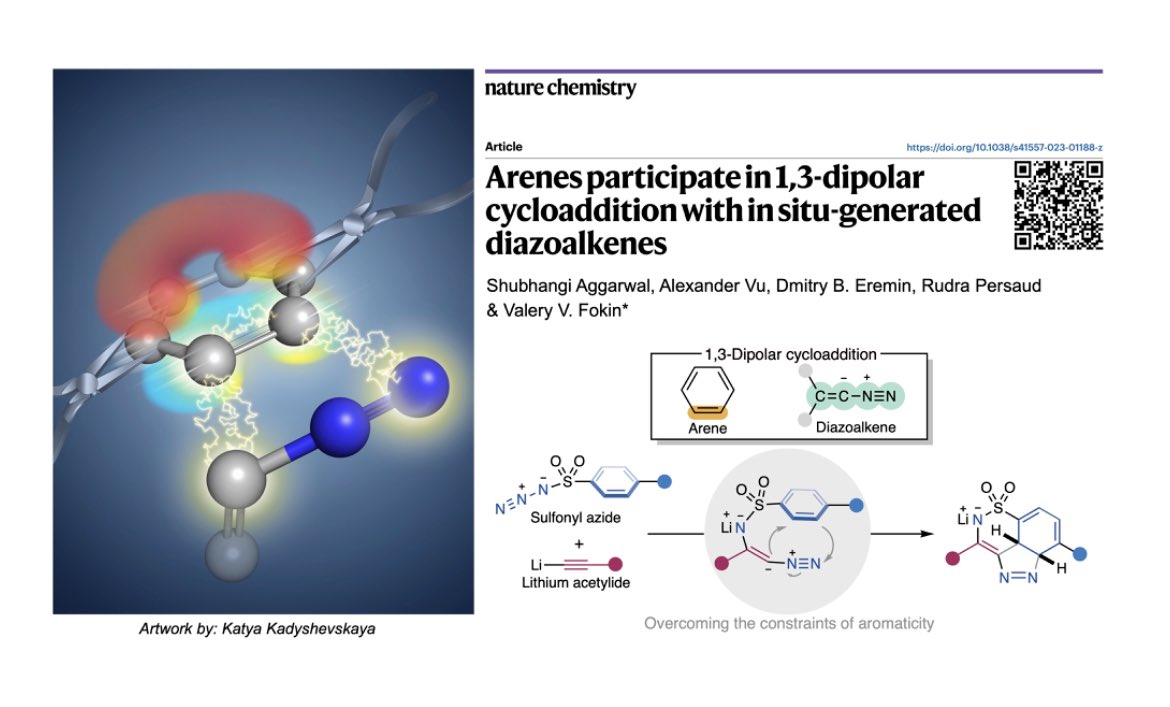
USC Bridge Institute (@USCBridge) / X

Arenes participate in 1,3-dipolar cycloaddition with in situ

纳米人

The Huisgen Reaction: Milestones of the 1,3‐Dipolar Cycloaddition

Facile (3+2) Cycloaddition between an N‐Heterocyclic Olefin and

1,3‐Dipolar Cycloaddition of Azomethine Ylides to Aldehydes

Molecules, Free Full-Text

The Truce–Smiles rearrangement and related reactions: a review

Synthesis of diazoolefin complexes The diazoolefin complexes 8–11

The Truce–Smiles rearrangement and related reactions: a review

Molecules, Free Full-Text