FDA approves over-the-counter sales of topical gel Eroxon for erectile dysfunction

The U.S. Food and Drug Administration has approved over-the-counter marketing for Eroxon, as a first-of-its-kind topical gel for erectile dysfunction.

FDA grants OTC authorization to topical gel for erectile dysfunction

Futura's novel ED formulation now available in Europe
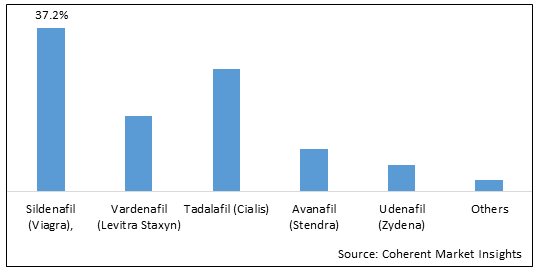
Erectile Dysfunction Market Size, Trends And Forecast To 2030

Futura Medical expects FDA approval for topical ED treatment during Q2

Topical Gel for Erectile Dysfunction Gets FDA Approval for Over-the-Counter Sale

World's First OTC Gel for Erectile Dysfunction, Eroxon, Erectile Dysfunction Treatment

MED3000 CLINICAL PROGRAMME - Futura Medical

Eroxon, First Over-The-Counter Erectile Dysfunction Gel, Gets FDA Approval

Eroxon StimGel Treatment Gel for Erectile Dysfunction - 4 Single Dose Tubes - 2 Pack : Health & Household

SMSNA - Over-the-Counter Gel for Treating Erectile Dysfunction Approved by FDA

Erectile dysfunction: Topical gel gets FDA nod for over-the-counter marketing

First-of-its-kind erectile dysfunction gel Eroxon gets FDA OK

Labatec Pharma launches Eroxon® in the UAE

Over-the-counter topical gel launched for erectile dysfunction - Trends in Urology & Men's Health - Wiley Online Library









