What Is Regulatory Dossier and What Does It Contain? - The Kolabtree Blog
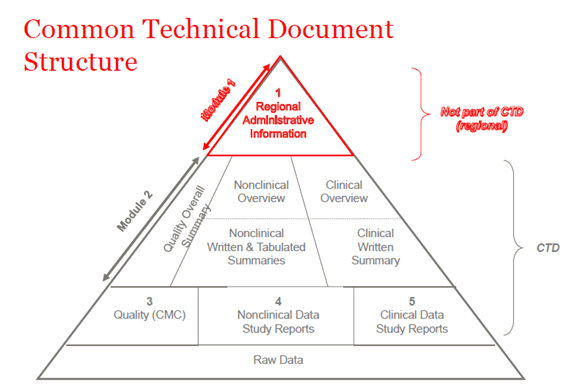

Regulatory Documents Freyr - Global Regulatory Solutions and Services Company

What Is Regulatory Dossier and What Does It Contain? - The Kolabtree Blog

Scientific and CTD Dossier Writing - BlueReg Group

7 Essential Skills to Look For When You Hire a Medical Writer

About the Author

Pharmaceutical Regulatory Services, CMC Writing

Clinical Trial Protocol - LifeProNow

Writing a Clinical Trial Protocol: Expert Tips - The Kolabtree Blog
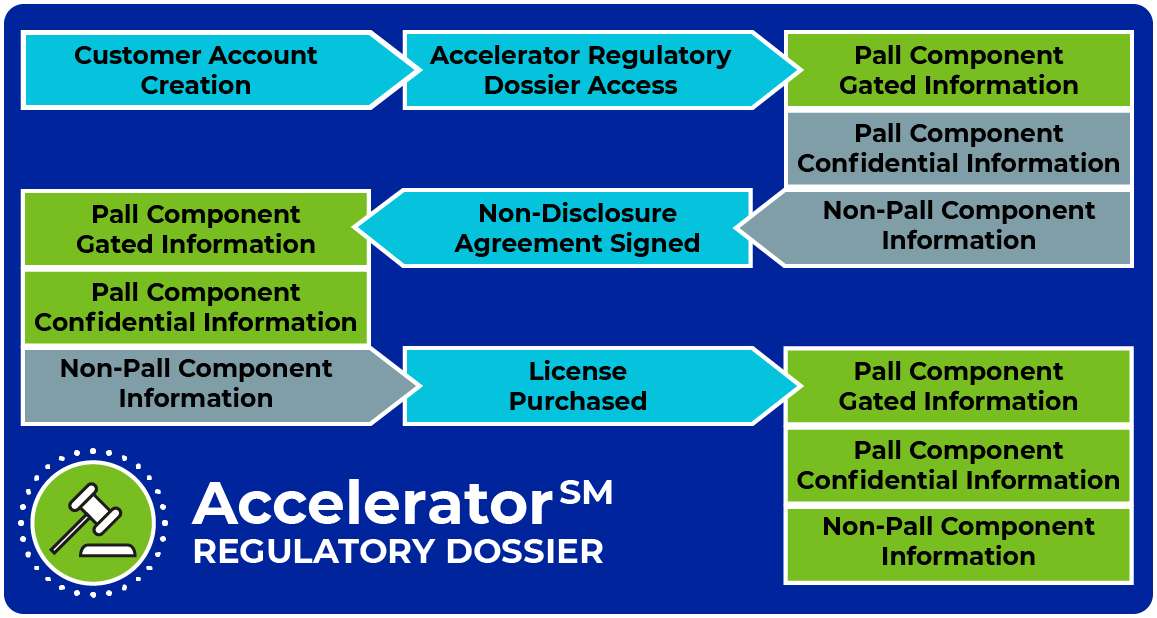
Biotech Regulatory Portal - Dossier

White Papers Freyr - Global Regulatory Solutions and Services Company
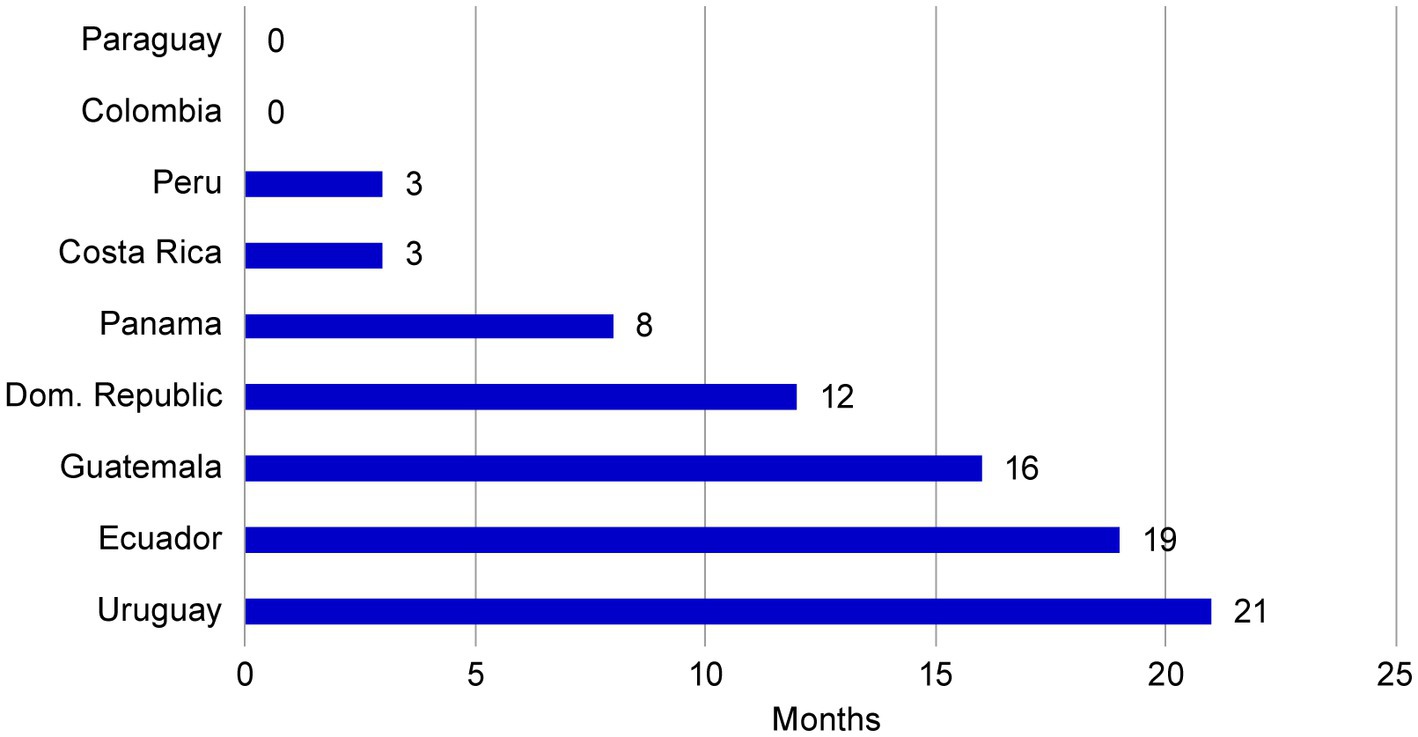
Frontiers Establishing a core dossier for multiple regulatory submissions: a case study in the Latin America region

Pharmaceutical Regulatory Services, CMC Writing

Drug development & compilation of registration dossier
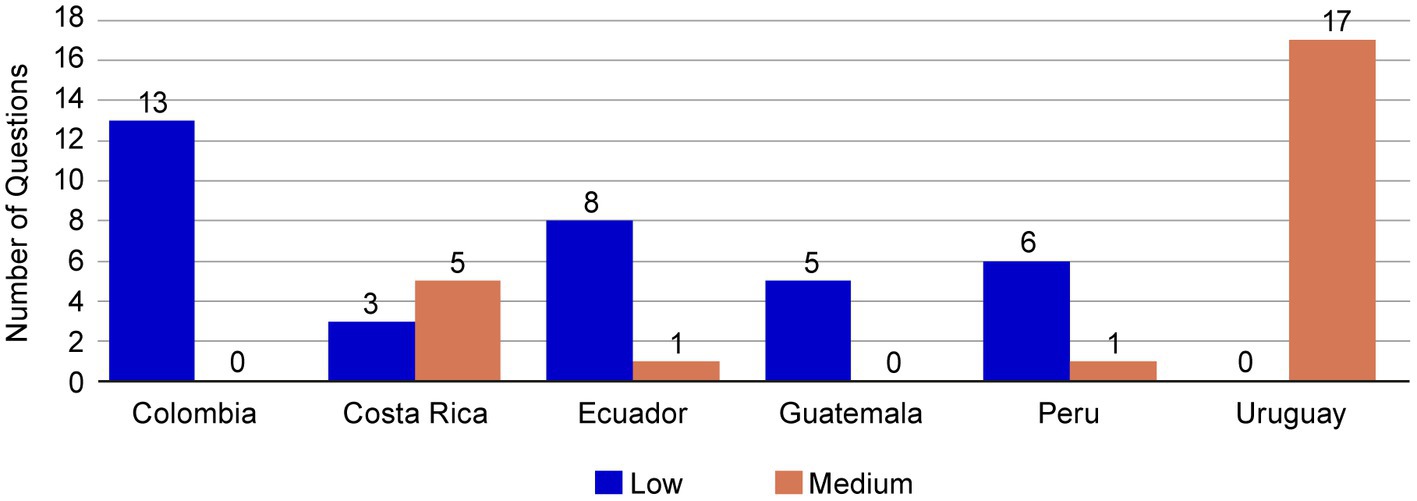
Frontiers Establishing a core dossier for multiple regulatory submissions: a case study in the Latin America region

Nare Simonyan - Freelance Regulatory Writing Consultant in Yerevan, Armenia









