CTL Amedica granted FDA 510(k) approval for Navigation Instrument

Medical Device Safety Service GmbH
Life Spine Gains FDA Clearance for Additions to ARx

Inline XBRL Viewer
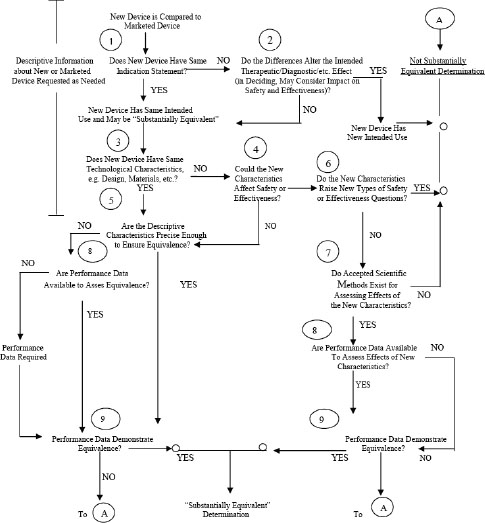
4 The 510(k) Clearance Process Medical Devices and the Public's
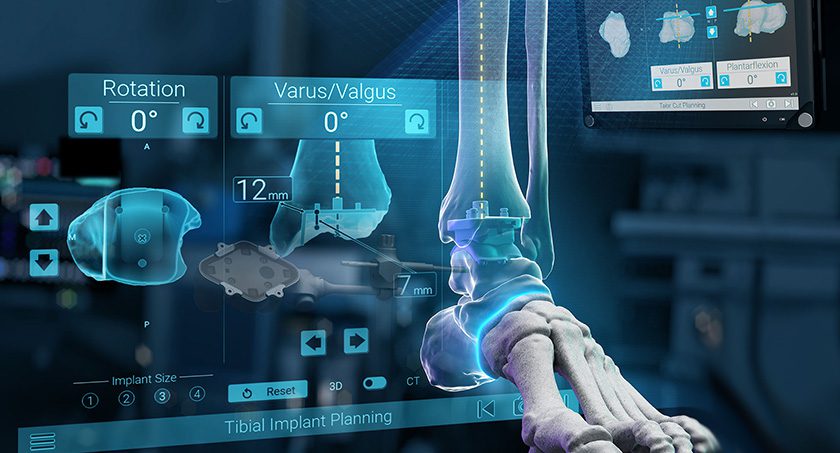
Fusion Robotics Gains 510(k) for Spinal Navigation & Robotics

FDA Regulation of Medical Devices and Software/Apps

Recent FDA 510(k) Clearances in Spine

Medical Device Testing Requirements for 510(k) Submissions - In

FDA's 510(K) Submission Process

FDA grants 510k clearance of the CTL Amedica Navigation Instrument

Medical Device Regulations - 510(k) Process

NEWS Archives - SPINEMarketGroup

2020 Archives - Page 6 of 16 - SPINEMarketGroup
%20predicate%20device.png?width=521&height=351&name=510(k)%20predicate%20device.png)
Everything you need to know about the FDA 510(k) submission